LEDs are one of the coolest electronic components. They can brighten up any circuits project and turn it in to an eye-catching bonanza of blinking lights. Okay, corny imagery aside, they’re pretty cool. But have you ever thought of how they actually work? You likely already know that LED stands for light emitting diode. Where does the light emit from, though? There are no bulbs or filaments in an LED. So what’s going on in there that produces the glow that we love to have in all of our circuits?
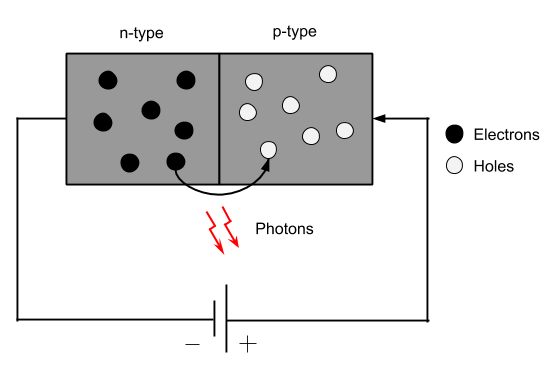
In LEDs, current flows from the anode to the cathode. If a voltage was applied to the cathode of the LED, nothing would happen. This operation is accomplished with a semiconductor that has what’s referred to as a p-n junction connected to the terminals of the LED. When a current is applied to the anode of the LED, it is the behavior of the aforementioned semiconductor that produces the light we see from the LED.
There are entire courses and textbooks dedicated to semiconductor technology, but this discussion is going to be quite brief. Recall from the previous paragraph that most LEDs utilize a semiconductor with a p-n junction. This refers to the fact that the semiconductor consists of n-type silicon joined to p-type silicon. N-type silicon is silicon with a few extra electrons and p-type silicon is silicon with a few missing electrons, referred to as holes. Holes can be thought of as the opposite of electrons (not to be confused with protons though). Generally, electrons seek to fill holes, and holes attract electrons to fill them. When a voltage is applied to the p-type side of the semiconductor, electrons and holes jump across the p-n junction, attempting to complete an electron/hole match. As an electron combines with a hole, an atom is completed and energy is released. This energy is released as a photon, the quantum (or smallest “unit”) of light. And this is what lights up an LED!