Hysteresis is something that is all around us. Its effects can be found in many disciplines, like economics and biology, but especially in engineering and physics. But what is it? How do we use it? Let’s do some digging to find out.
According to Merriam-Webster, hysteresis is defined as “a retardation of an effect when the forces acting upon a body are changed.” Also “the lagging of a physical effect on a body behind its cause.”
Wikipedia gives a slightly different definition: “the dependence of the output of a system not only on its current input, but also on its history of past inputs.”
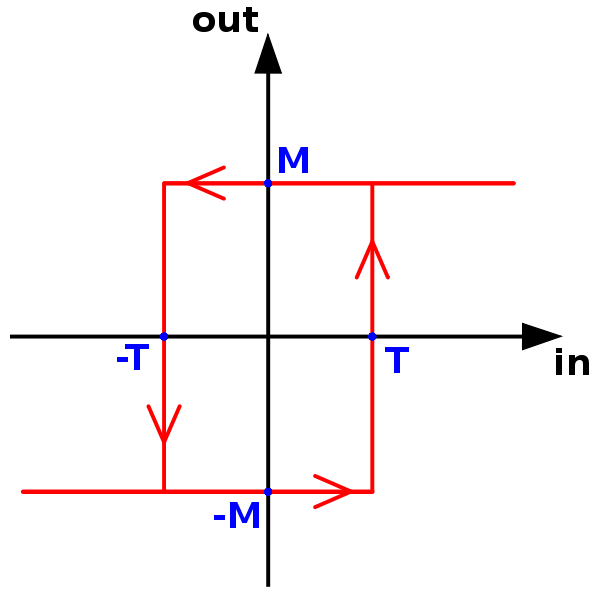
So what is it really then? I think the best way to explain it is with some examples.
We all know that a piece of iron metal, like a nail, can be magnetized by the use of an appropriate force. Once it is magnetized it stays that way, but it can be demagnetized as well if an appropriate opposing force is applied. It changes from one state to the other by placing it in a magnetic field. But doing so doesn’t effect an immediate change on the nail. It takes time for the atomic dipoles to change their orientation to align with the magnetic field. If you remove the nail before the dipoles have finished moving, it won’t be fully magnetized if at all. If you leave it long enough, the nail is magnetized and won’t change unless you place it in a magnetic field with opposing orientation (just flip it end to end). You can’t magnetize a nail without an outside force or if it is already magnetized, and you can’t demagnetize it without an outside force or if it is already demagnetized. Hysteresis.
Another example is the solid/liquid phase transition of a material called agar, which is derived from algae. It is commonly used as an ingredient in Asian desserts and as the substrate in petri dishes for microbiological studies. It’s also used in ant farms as a sand substitute and nutrient source because it is clear and allows for open observation of the colony. It solidifies between 32° and 40°C, but melts at 85°C. This means that between 40° and 85°C, its present phase depends on whether it was previously a solid or a liquid. If you melt it at 85°C and then cool it, it will be a liquid when cooled to 60°C. But if it is solid at 40°C, it will remain a solid when raised to 60°C. Weird, I know –but that’s hysteresis.
One last example is the thermostat in your home. You set it at whatever is comfortable, let’s say 20°C (68°F) and then walk away. The system will sense the current temperature and then turn it on or off depending on whether it needs to increase or decrease the room temperature. Let’s say it starts at 15°C. It turns on and starts to warm up. Once it hits 20°C, it doesn’t immediately turn off, but keeps running for a short time and raises the temperature to 21°C instead. It then shuts off and the room starts to cool. Once it hits 20°C again, it doesn’t immediately turn back on, but instead waits until the room has cooled another degree to 19°C. The system then turns on and the cycle starts again. There is a lag in response to the ambient temperature which has been programmed into the system. This keeps the system from switching on and off rapidly around that 20°C sweet spot.
So how can we use it? Finite state machines are probably the easiest and most obvious example. They control a large percentage of things around us that depend on knowing the past state of the system to determine what to do next. Your thermostat may well use sequential logic as a state machine, but a vending machine is probably the simplest example. As you add coins, it keeps track of how much has been added and then spits out your selection once the threshold is reached.
Hysteresis is something we deal with everyday. It controls when your turn signal indicator turns off, when the vending machine spits out your chocolate fix, whether the digital lock code unlocks your safe or not, and the fact that your computer hard drive even functions. It’s everywhere, and thank goodness for it.

